One-stop Technology Platform to Accelerate Drug Discovery and Development
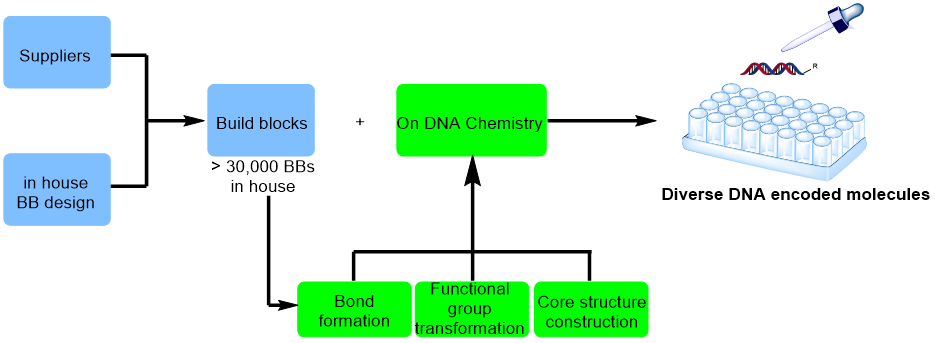
The most important advantage of DELT is to enable the exploration of huge chemical space in a cost-effective and timely manner. The diversity of chemical structures in the libraries is one of the key properties of DELs. While DELs are synthesized by combinational assembly of building blocks (BBs) using a spilt-pool strategy in aqueous solution, multitudinous BBs and DNA-compatible chemistries are fundamental for constructing diverse and drug-like compounds in DELs. HitGen has made great efforts to expand the chemistry toolbox for DEL synthesis.
There are over 30,000 BBs at HitGen selected from over 100,000 commercially available chemicals by considering structural diversity, pharmacophore representation, chemical reactivity and toxophore filtration. Most of those are mono-functional chemicals such as amines, acids, aldehyde, etc. Also there are bi-functional chemical and tri-functional chemicals collected from commercial sources or designed and synthesized by a dedicated team at HitGen which could improve the diversity and novelty of DEL products.
DNA-compatible chemistry plays a key role in DEL quality. Since DELs are synthesized by combinational assembly of BBs in the presence of DNA tag, the reactions have to be performed in aqueous phase with good solubility. With the advancement of synthetic chemistry, enormous bond formation reactions could be applied to construct novel structures flexibly and conveniently by medicinal chemists. To explore the chemical space with more drug-like molecules from HitGen DELs, on-DNA chemistry R&D was carried out with bond formation, functional group (FG) transformation, core structure construction, etc. With persistent effort, more than 100 types of protocol for bond formation, heterocycle in-situ generation, functional group transformation and other application have been established in HitGen's DEL platform.
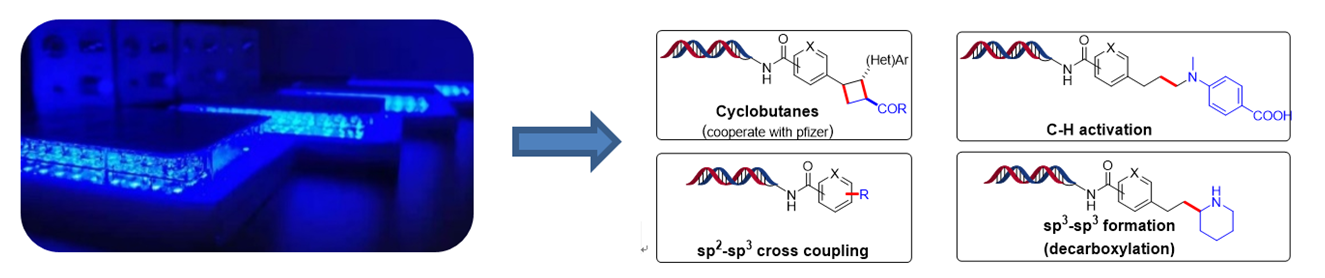
On-DNA bond formation
With the advancement of synthetic chemistry, bond formation reactions are widely applied by medicinal chemists to construct novel structures. However, many of reactions are difficult to achieve during DEL construction since it is greatly limited by DNA stability and solubility, namely DNA-compatability issue. With great efforts from chemists, multiple on-DNA bond-formation chemistries have been made successfully, such as C-C, C-N, C-O, C-S, etc. It is well known that photoredox catalysis is now an indispensable tool in organic synthesis due to its mild condition and wide substrate scope. Nowadays, various photoredox transformations including C-H activation, alkene addition, cross-coupling, etc., have been developed and applied in our DEL construction. In 2020, as part of collaboration with Pfizer, the photoredox catalyzed [2+2] cyclization was published on Organic Letters (Kölmel, D. K.; Ratnayake, A. S.; Flanagan, M. E.; Tsai, M-H.; Duan, C.; Song, C. Org. Lett. 2020, 22, 2908–2913).
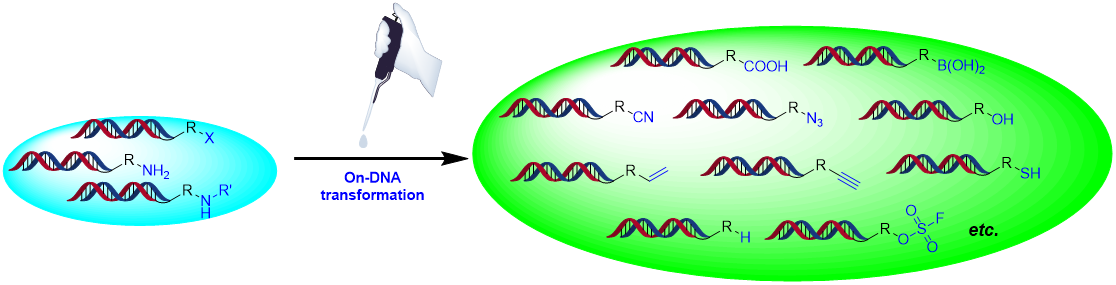
On-DNA functional group transformation
Functional groups are critical for new bond formation which serve as an indispensable handle in chemical transformations. On-DNA functional group transformation development can enrich the sources of structure with multi-functional groups. To expand the structure diversity, HitGen has established at least 20 functional group transformation/swap protocols which expand the chemical transformations for limited functional groups of commercially available reagents.
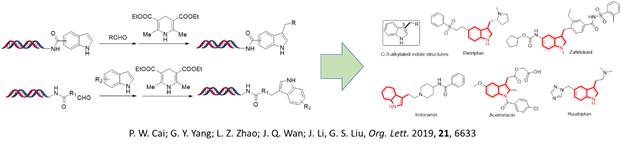
On-DNA core structures construction
Privileged structures are of great interest for medicinal chemistry. HitGen’s scientists have successfully developed on-DNA chemistry to include key privileged core structures of drugs into DELs, such as C3-alkylated indoles (Org. Lett. 2019, 21, 6633–6637), 2-aminobenzimidazole (Org. Lett. 2020, 22, 1290–1294), Indazolone (Org. Lett. 2020, 22, 16, 6277–6282), substituted pyrimidine (Chem Asian J. 2020, 15, 1–6), etc. Out of the top 100 privileged structures to appear in FDA approved drugs, up to ninety have been covered in our DEL products.
Beyond aqueous systems for on-DNA chemistry development
DNA-encoded libraries have shown promise as a valuable technology for enabling the hit discovery process. Although DEL provides relatively inexpensive access to libraries of unprecedented size, their production has been hampered by the needs of the encoding DNA tags with DEL-compatible chemistry to dilute aqueous environments. Recently, Reversible Adsorption to Solid Support (RASS) have been demonstrated as promising methods to expand DEL reactivity and application scenarios (Angew. Chem. Int. Ed. 2020, 59, 19, 7377-7383). Many efforts have been dedicated into the above innovation at HitGen. It’s our view that DELT has already shown great promise in the generation of novel chemical leads, and has considerable scope to develop in terms of molecule types and the way these molecules are produced in the future.
We use cookies to provide a better web experience.
By using our site, you acknowledge our use of cookies and please read our Cookie Notice for
More information