One-stop Technology Platform to Accelerate Drug Discovery and Development
HitGen has the in-house expertise and automatic equipment to undertake high-throughput screening of thousands of crystallization conditions, and through world-class synchrotron facilities, such as the SSRF (Shanghai, China), and the Spring-8 synchrotron radiation facility, (Hyogo, Japan), we can collect high-resolution structural datasets for apo or ligand-bound target proteins. The data are processed by our experts, and the solved the structures will be presented in a variety of formats suitable for computational modelling.
General crystallography process
Step1: Crystallization screening (to identify the conditions for crystal growth)
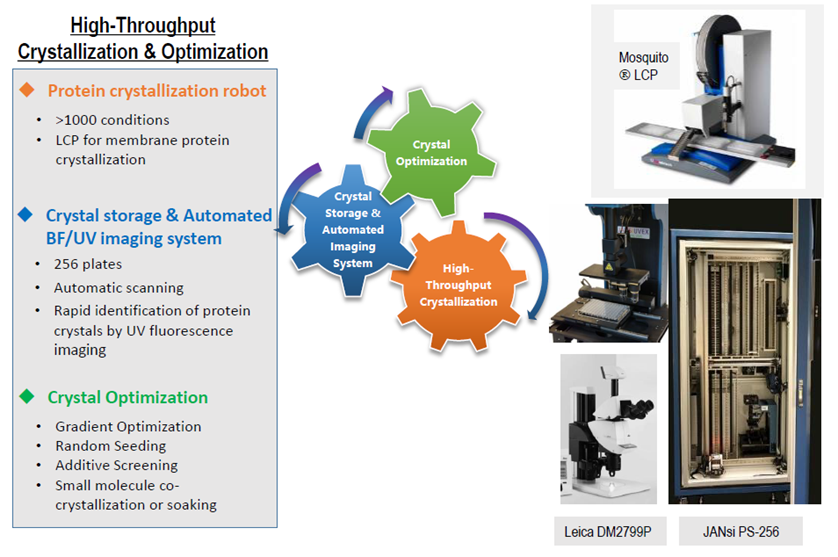
●Initial screening will be set up to 1000-2000 conditions at different temperatures, using our high throughput nanoliter dispensing robot (Mosquito ÒLCP), saving time and making efficient use of precious samples.
●Automated imaging for crystal evaluation (UVEX-ps256, Jansi)
●Daily diffraction experiment is performed on home lab X-ray (Bruker D8 VENTURE with METAJET and CPAD)
Step2: Crystallization optimization (by multi-dimensions for optimal crystal conditions)
●Correlation of images & experimental setup (database)
●Reproduction and multi-dimensional optimization of crystal conditions
●Crystals from coarse matrix screens are optimized using fine screens, additives, and seeding.
Step 3: Crystal Structure Determination
Co-crystallization with ligands (mainly small molecule compounds from DEL screening or clients’ compounds) or soaking them in the apo protein crystals will be performed prior to data collection;
X-ray data collection is mainly performed on a monthly schedule at the industrial synchrotron beamlines (SSRF or Spring-8), ensuring the highest resolution for the crystal structure.
We use cookies to provide a better web experience.
By using our site, you acknowledge our use of cookies and please read our Cookie Notice for
More information