One-stop Technology Platform to Accelerate Drug Discovery and Development
Protein-protein interactions (PPI) are physical and chemical contacts between two or more protein molecules. As a fundamental aspect of almost all biological processes, any interference of the sophisticated PPI network could result in potential physiological disorder or disease. Although some PPI networks have been well-established for their roles in tumor development and therefore have been recognized as potential oncological targets (Figure 1), PPI-based drug discovery is challenging due to its possible event-driven conformational alteration and interface accessibility variation. DNA-encoded Library (DEL) selection, an affinity-based small molecule selection process, is considered a powerful PPI-based drug discovery tool. With strong understanding of the target PPI mechanism of actions (MOAs) and rational design of interaction partner group, DEL is capable of providing selective hits with desirable MOA and potentially, designated function.
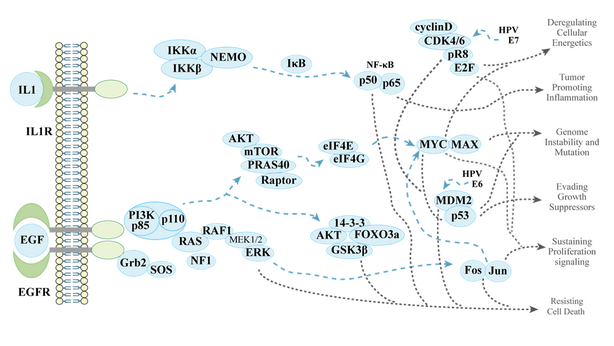
Figure 1, an example of PPI network in tumor development related processes.
Direct PPI blocker identification
One of the biggest challenges for PPI disruptor identification is that the “coverage” of small molecules might differ from that of the interacting motifs in PPI process. This is largely due to the aforementioned intrinsic nature of PPI that the interaction sites are more of a shallow and large interface rather than a deep and well-defined pocket. With its great diversity, DEL has been providing PPI interface binders for over a decade at HitGen, and a large portion of the identified binders has been later confirmed as bona fide PPI blockers in the follow-up validation studies. For example, aiming to target the interactions between the IL17A and IL17RA for treatment of autoimmune and autoinflammatory diseases, HitGen has conducted a DEL selection, and novel IL17A/IL17RA PPI interrupter have been identified and compounds through medicinal chemistry optimization have now been nominated as preclinical candidates.
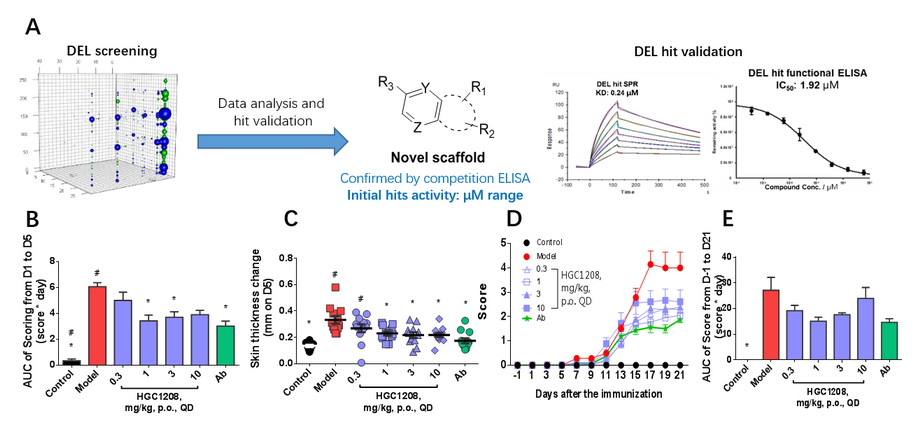
Figure 2, A) A successful hit series identified by DEL for direct IL17A/IL17RA interaction disruption. DEL-derived compounds showed efficacy in IMQ-Induced psoriasis model (B, C) and EAE model (D, E).
“Smart Selection Campaign”: function oriented hit identification
PPI could be modulated due to the conformational alterations of the interacting proteins upon particular signaling event, such as ligand binding, co-factor interaction, pH variation, and others. These PPI regulation processes could lead to pathway activations upon complex formation, or switches to be turned on and off with domain orientation change upon presence or absence of co-factors. At HitGen, “Smart Selection Campaign” was developed to explore the binding capability alteration and conformation-based regulation in PPI ligand discovery. By conducting DEL screening in the absence or presence of partner proteins, co-factors, activators and/or inhibitors, this hit identification process has already provided vast amount of information of potential function of the candidate.
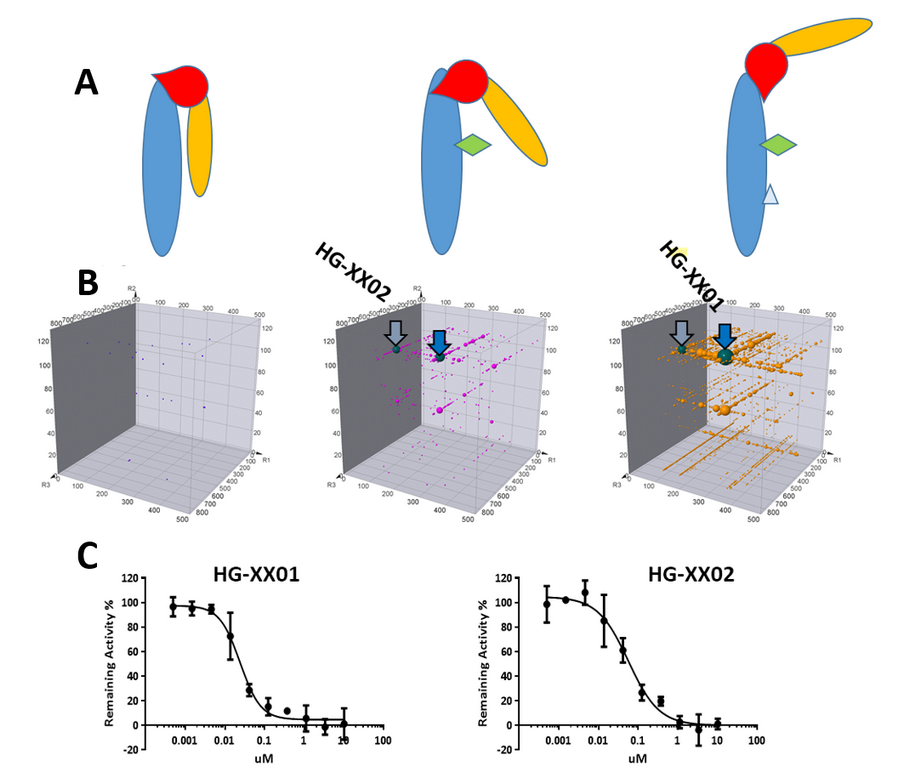
Figure 3, A) Conformation change upon presence of different cofactors for a PPI target introduces different binding capabilities. B) Different series of compounds identified from DEL selection with conformational specificity. C) Confirmed hits from different series.
Identification of specific binder with Special MOA
In a particular DEL selection conducted at HitGen, the PPI network for the target of interest was complicated because different pathways are activated upon binding to different co-factors. On the one hand, target protein could form a heterodimer with partner protein 1 and further form a ternary complex with protein 2 to initiate the downstream signaling pathway. On the other hand, the same target protein could form a different heterodimer with partner protein 3, leading to a bypass pathway. By including all the partner proteins in our DEL selection with different combinations to reflect all possible scenarios, several hit series were identified, and one of the series could specifically bind to the target-protein 1 complex. In the subsequent hit validation studies, we also found that this binder series could stabilize the target-protien1 complex and more importantly, this stabilization prevents the formation of either the ternary complex, or the target-protein 3 complex. Therefore, we have identified a specific PPI stabilizer series with dual functions at once.
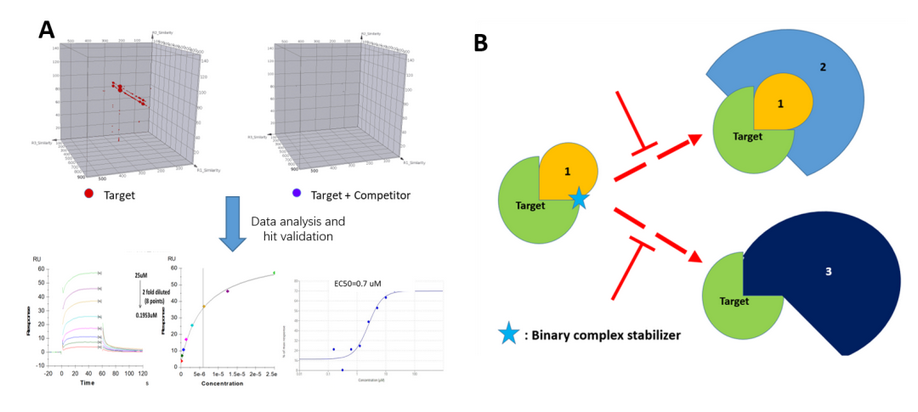
Figure 4, A) a series of hits identified by DEL and confirmed with orthogonal methods. B) Further MOA study demonstrated that in the presence of the confirmed hit compound, two PPI cascades have been interfered based on a special MOA.
DEL for PPI at HitGen
With profound understanding of the PPI target MOA and highly case-dependent DEL selection plan, all possible conformational alterations and different interactions with different partners will be included in our selection to simulate the actual PPI processes and maximize the chance of identifying robust hits with good affinities and properties. HitGen’s “Smart Selection Campaign” (Figure 5) along with our trillion size DEL, have enabled PPI-targeted drug discovery with easier design and more straightforward execution.
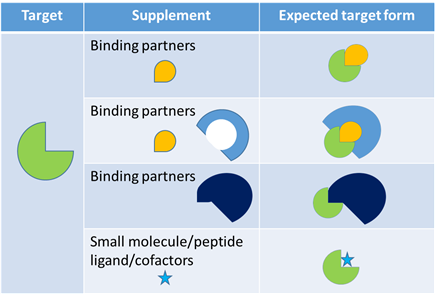
Figure 5, An example of possible selection groups to explore all possible target forms in one selection campaign
We use cookies to provide a better web experience.
By using our site, you acknowledge our use of cookies and please read our Cookie Notice for
More information