One-stop Technology Platform to Accelerate Drug Discovery and Development
HitGen recognizes application of biophysical methods and technologies an important role in different stages of drug discovery and development, especially during hit validation process. Robust improvement in methodology and instrumentation makes rapid and high throughput biomolecular interaction characterization achievable at HitGen with unprecedented quality. By establishing a comprehensive biophysical testing platform, HitGen is capable of providing accurate, real-time, and label-free interaction analysis data including both affinity and kinetic information, i.e. association rate constant (Ka), dissociation rate constant (Kd), dissociation constant (KD), entropy/enthalpy change regarding intermolecular interactions.
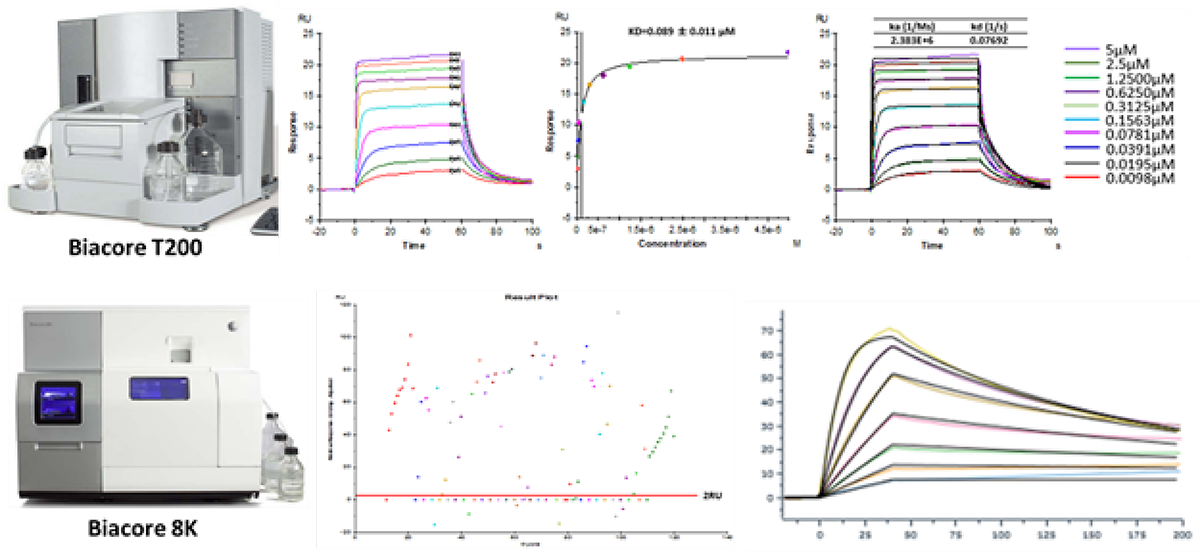
Surface plasmon resonance (SPR)
Cutting-edge Biacore™ series SPR platform has become a rapid, highly-sensitive, and label-free tool for hit validation as well as hit-to-lead optimization.
Biacore™ T200 and Biacore™ 8K guaranteed efficient and accurate data generation with minimized hassle and maximized throughput.
The platform offers both affinity and kinetic information with thorough analysis in a single run.
Comprehensive understanding of biophysics and methodology enabled HitGen to offer not only precise interpretation of the data but also customized guidance for hit optimization and drug discovery.
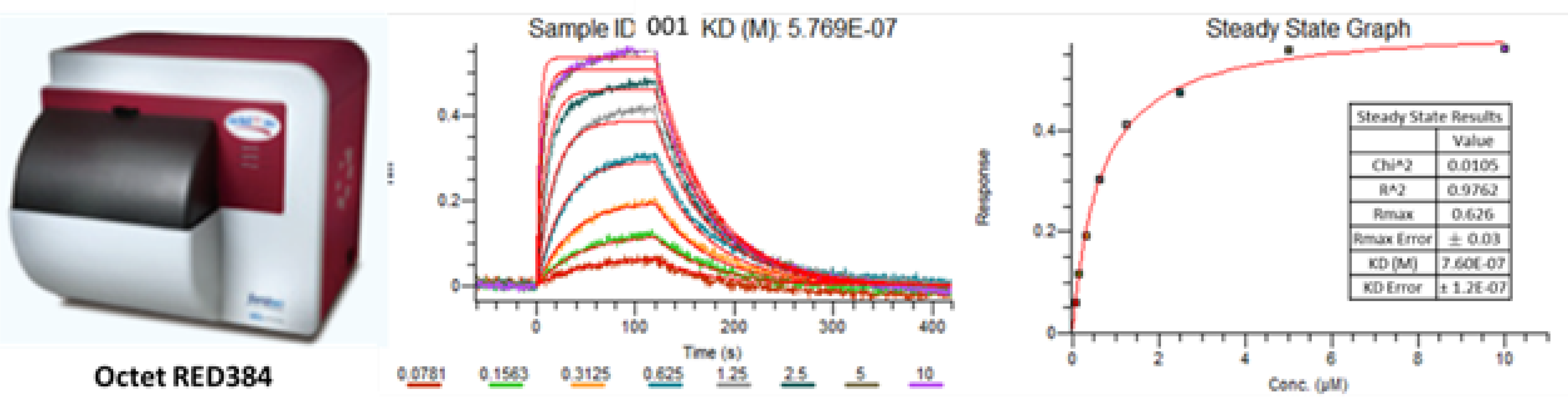
BioLayer interferometry (BLI)
BioLayer interferometry (BLI), as an optical analytical technique, is capable of measuring kinetics and affinity of biomolecular interactions in a real-time fashion.
Octet RED384 System is designed for the analysis of biomolecular interactions in real time in the determination of kinetic constants.
With sixteen biosensors, Octet allows a variety of conditions to be monitored, and compared in real-time. This enables multiple concentrations, different antibodies, epitopes, clones, inhibitors, controls or different media types, and multiple layers of protein binding to be studied and monitored simultaneously.
Highly sensitive and high capability of Octet facilitates hit validation process and therefore shortened the timeline for novel drug discovery.
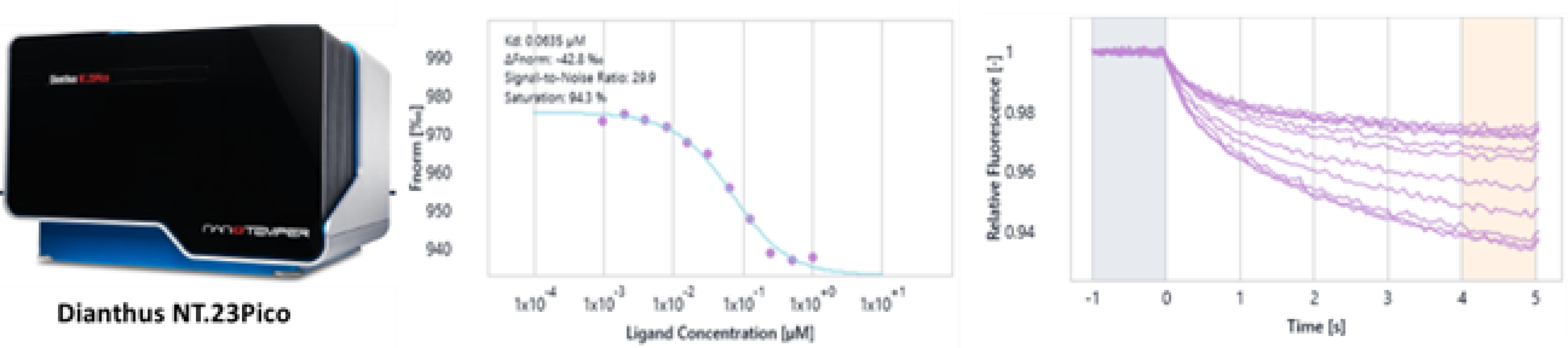
Temperature related intensity change (TRIC)
Temperature related intensity change (TRIC) based Dianthus NT.23Pico, by measuring a fluorescence intensity change upon a precise laser induced heat, provides fast decision-making on the right candidates and acceleration on lead optimization.
Dianthus NT.23Pico generates easy-to-interpret affinity ranking tables and histograms with great capacity: a full 384-plate containing different ligands and targets can be monitored and analyzed within 1 hour.
There is little to no limitation of the sample form, which makes it possible to analyze aqueous solution, plasma, and cell lysate.
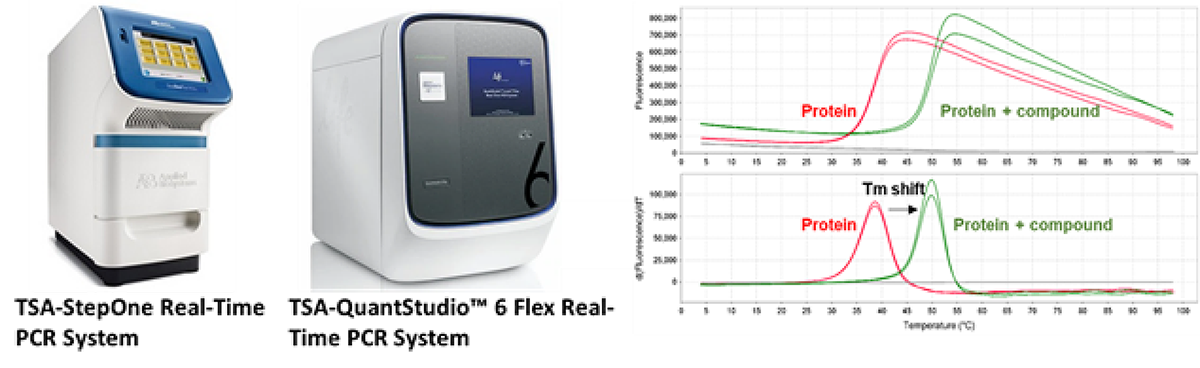
Thermal shift assay (TSA)
Melting point (Tm) shift measures thermal stability of target protein under various conditions and therefore offers a quick method to determine the binding between target protein and ligand.
TSA monitors the shift of Tm at the presence of ligand, the result will indicate whether the ligand would stabilized or de-stabilize the target.
HitGen equips both 96-well and 384-well capable real-time PCR systems which is capable of high-throughput TSA study.
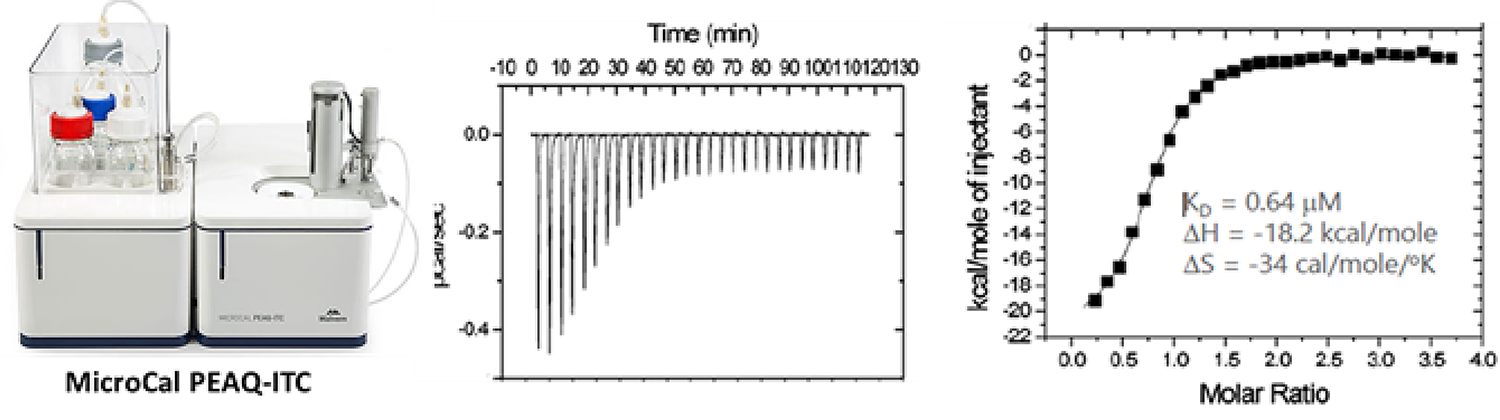
Isothermal titration calorimeter (ITC)
By directly measuring heat absorbed or released during the binding event, isothermal titration calorimeter (ITC) provides accurate reads of binding parameters including binding affinity (KD), stoichiometry (n), enthalpy (ΔH), and entropy (ΔS) in a single experiment.
HitGen has equipped MicroCal PEAQ-ITC, which enabled in-solution biomolecular interaction with high sensitivity, low volume, and free of labeling.
We use cookies to provide a better web experience.
By using our site, you acknowledge our use of cookies and please read our Cookie Notice for
More information